

Sharon Hammes-Schiffer
Contact:
Sharon Hammes-Schiffer
A. Barton Hepburn Professor of Chemistry
[email protected]
Frick Laboratory, 353
609-258-4986
Faculty Assistant:
Julia Wlodarczyk-Mingozzi
Faculty/Grants Assistant
[email protected]
Frick Laboratory, 389
609-258-3674
Research Focus
Our research centers on the development and application of theoretical and computational methods to understand the fundamental physical principles underlying chemical processes. One of our main interests is proton-coupled electron transfer (PCET) reactions, which involve the coupled motions of electrons and protons and play a critical role throughout chemistry and biology. More generally, our research encompasses charge transfer and catalysis in molecular, electrochemical, photochemical, and biological systems.
Our group has developed a general theoretical formulation for PCET and is continuing to extend this theory to investigate important chemical and biological systems. This theory treats the electrons and transferring protons quantum mechanically, including both electron and hydrogen tunneling, and incorporates the motions of the proton donor-acceptor mode and the solvent or protein environment. Analytical rate constant expressions have been derived in various well-defined regimes, enabling the calculation of rate constants and kinetic isotope effects for comparison to experiment. In addition to developing PCET theories, our group works closely with experimental collaborators in a wide range of applications spanning molecular and heterogeneous catalysis, electrochemistry, photochemistry, enzymology, and photoreceptors.
Another active area is the development of theoretical methods for including nuclear quantum effects, such as zero-point energy and hydrogen tunneling, in quantum chemistry calculations and molecular dynamics simulations. Our group originally developed the nuclear-electronic orbital (NEO) method and is continuing to explore new directions within the NEO framework. In the NEO approach, specified nuclei are treated quantum mechanically on the same level as the electrons using density functional theory or wavefunction theories without invoking the Born-Oppenheimer separation between the electrons and the quantum nuclei. This approach allows real-time nuclear-electronic quantum dynamical simulations of processes such as photoinduced proton transfer, PCET, molecular polaritons, and plasmon-induced chemical reactions. The NEO methods are being incorporated into many of the popular quantum chemistry software packages.
Our group uses a combination of these types of analytical theories and computational methods to investigate fundamental aspects of chemical processes. The diverse applications include molecular electrocatalysts, proton wires, artificial photosynthetic systems, nanoparticles, heterogeneous electrochemical systems, enzymes, and photoreceptor proteins. Our simulations of these systems have elucidated the critical roles of hydrogen tunneling, electrostatics, reorganization, and conformational motions.
Honors
- Willard Gibbs Medal Award, 2021
- Joseph O. Hirschfelder Prize in Theoretical Chemistry, 2021
- American Chemical Society Award in Theoretical Chemistry, 2021
- Royal Society of Chemistry Bourke Award, 2020
- G. M. Kosolapoff Award, 2019
- Fellow, Biophysical Society, 2015
- Member, International Academy of Quantum Molecular Science, 2014
- Member, U.S. National Academy of Sciences, 2013
- Fellow, American Association for the Advancement of Science, 2013
- Member, American Academy of Arts and Sciences, 2012
- Fellow, American Chemical Society, 2011
- Fellow, American Physical Society, 2010
- American Chemical Society Akron Section Award, 2008
- International Academy of Quantum Molecular Science Medal, 2005
- Iota Sigma Pi Agnes Fay Morgan Research Award, 2005
- Camille Dreyfus Teacher-Scholar Award, 1999
- Alfred P. Sloan Research Fellowship, 1998
- National Science Foundation CAREER Award, 1996
Selected Publications
S. Hammes-Schiffer, “Exploring proton-coupled electron transfer at multiple scales.” Nat. Comp. Sci. 2023, 3, 291-300.
T. E. Li and S. Hammes-Schiffer, “Nuclear-electronic orbital quantum dynamics of plasmon-driven H2 photodissociation.” J. Am. Chem. Soc. 2023, 145, 18210-18214.
J. Xu, R. Zhou, V. Blum, T. E. Li, S. Hammes-Schiffer, and Y. Kanai, “First-principles approach for coupled quantum dynamics of electrons and protons in heterogeneous systems.” Phys. Rev. Lett. 2023, 131, 238002.
W. R. Lake, J. Meng, J. M. Dawlaty, T. Lian, and S. Hammes-Schiffer, “Electro-inductive effect dominates vibrational frequency shifts of conjugated probes on gold electrodes.” J. Am. Chem. Soc. 2023, 145, 22548-22554.
K. Cui, A. V. Soudackov, M. C. Kessinger, J. Xu, G. J. Meyer, and S. Hammes-Schiffer, “General kinetic model for pH dependence of proton-coupled electron transfer: Application to an electrochemical water oxidation system.” J. Am. Chem. Soc. 2023, 145, 19321-29332.
J. Zhong, C. R. Reinhardt, and S. Hammes-Schiffer, “Direct proton-coupled electron transfer between interfacial tyrosines in ribonucleotide reductase.” J. Am. Chem. Soc. 2023, 145, 4784-4790.
M. Chow, T. E. Li, and S. Hammes-Schiffer, “Nuclear-electronic orbital quantum mechanical/molecular mechanical real-time dynamics.” J. Phys. Chem. Lett. 2023, 14, 9556-9562.
T. E. Li, Z. Tao, and S. Hammes-Schiffer, “Semiclassical real-time nuclear-electronic orbital approach for molecular polaritons: Unified theory of electronic and vibrational strong couplings.” J. Chem. Theory Comp. 2022, 18, 2774-2784.
Q. Yu, S. Roy, and S. Hammes-Schiffer, “Nonadiabatic dynamics of hydrogen tunneling with nuclear-electronic orbital multistate density functional theory.” J. Chem. Theory Comput. 2022, 18, 7132-7141.
B. P. Rimgard, Z. Tao, G. A. Parada, L. F. Cotter, S. Hammes-Schiffer, J. M. Mayer, and L. Hammarström, “Proton-coupled energy transfer in molecular triads.” Science 2022, 377, 742-747.
S. Hammes-Schiffer, “Nuclear-electronic orbital methods: Foundations and prospects.” J. Chem. Phys. 2021, 155, 030901.
R. E. Warburton, P. Hutchison, M. N. Jackson, M. L. Pegis, Y. Surendranath, and S. Hammes-Schiffer, “Interfacial field-driven proton-coupled electron transfer at graphite-conjugated organic acids.” J. Am. Chem. Soc. 2020, 142, 20855-20864.
J. J. Goings, P. Li, Q. Zhu, and S. Hammes-Schiffer, “Formation of an unusual glutamine tautomer in a blue-light using flavin photocycle characterizes the light-adapted state.” Proc. Nat. Acad. Sci. USA 2020, 117, 26626-26632.
E. Odella, B. L. Wadsworth, S. J. Mora, J. J. Goings, M. T. Huynh, D. Gust, T. A. Moore, G. F. Moore, S. Hammes-Schiffer, and A. L. Moore, “Proton-coupled electron transfer drives long-range proton translocation in bioinspired systems.” J. Am. Chem. Soc. 2019, 141, 14057-14061.
G. A. Parada, Z. K. Goldsmith, S. Kolmar, B. P. Rimgard, B. Q. Mercado, L. Hammarström, S. Hammes-Schiffer, and J. M. Mayer, “Concerted proton-electron transfer reactions in the Marcus inverted region.” Science 2019, 364, 471-475.
Related News
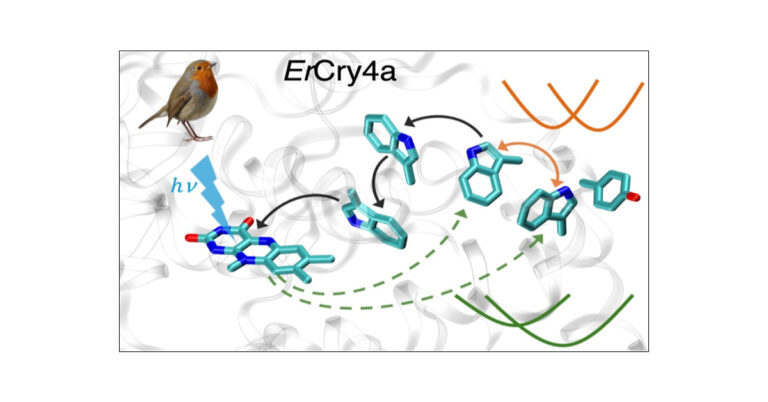
Collaboration provides computational support for cryptochrome-based magnetoreception in birds


