Electronic Excitations Transform Semimetal
In an investigation that may inspire new ideas about the application of light on ultrafast timescales to study material characteristics, researchers in the Schoop Lab and the Scholes Group are reporting results that could provide evidence for a rare photo-induced phase transition in a topological semimetal.
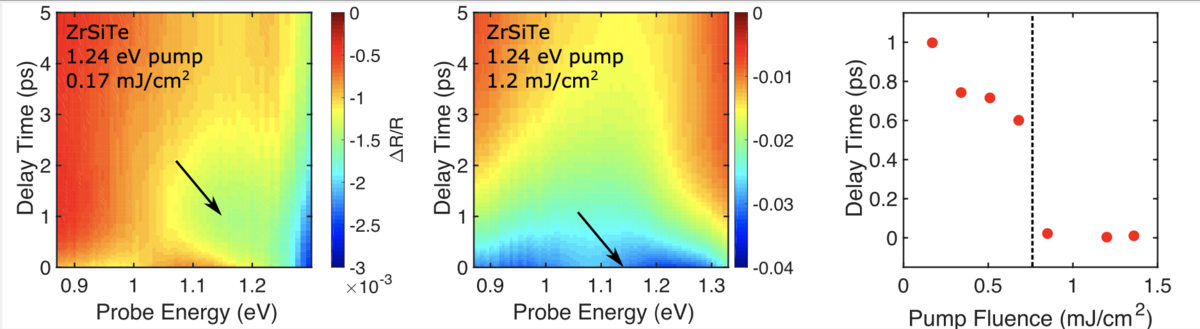
In their investigation, researchers found that the injection of electronic excitations dynamically modified the material, zirconium silicon telluride (ZrSiTe), in what they speculate is the signature of a Lifshitz Transition. A Lifshitz Transition (LT) is a type of phase transition in which the material’s Fermi surface abruptly changes but does not experience an accompanying symmetry change in the crystal structure, as with most such phase transitions.

Photo by C. Todd Reichart
Researchers used pulses of light on ultrafast timescales – deployed in the trillionths of a second – to induce the electronic phase transition in the material sample. If proven, the investigation could reveal ZrSiTe as an ideal candidate for high-frequency optoelectronic devices.
Typically, phase transitions like this require either very high or low temperatures or hundreds of thousands of atmospheres worth of pressure applied to the sample. Yet here, using lasers in ambient temperature conditions, researchers believe they triggered the transition without the usual inducements.
In addition, they report that the transitions don’t last long – maybe a few picoseconds or nanoseconds – and that they’re reversible.
The research could demonstrate ZrSiTe as a potential system from which to gain a better understanding of an exciting class of ultrafast, photo-induced phase transitions.
Further experiments to confirm the speculative results will be performed at a laboratory in Trieste, Italy later this year as soon as international travel becomes possible.
“Until maybe last year, these transformations have only ever been seen to exist in equilibrium. So, if you changed the temperature, you might see a Lifshitz Transition in certain materials,” said Robert Kirby, a fourth-year graduate student working jointly in the Schoop Lab and the Scholes Group. He is lead author on the research paper.
“But people haven’t been able to use light to make materials go through this kind of transition. As far as we’re aware – if what we see is correct – then this would be the second material ever to be shown to be capable of undergoing a photo-inducted LT at ambient temperatures,” he added.
“We definitely weren’t expecting to see this sort of phase transformation behavior in this material. ‘Why’ is definitely a good question. Why this material and not others? We’ve got more experiments to run to show whether what we hypothesized in this paper is what we think it is.”
Graphic courtesy of Robert Kirby
The paper, “Signature of an Ultrafast Photo-Induced Lifshitz Transition in the Nodal-Line Semimetal ZrSiTe,” appeared last week in Physical Review B. The work was primarily a collaboration between the Schoop Lab and the Scholes Group here at Princeton’s Department of Chemistry.
“Our study also shows how much you can achieve with interdisciplinary efforts,” said Assistant Professor of Chemistry Leslie Schoop. “We were able to find this phenomenon by combining optics and materials science here in our department.”
Colleagues at the Quantum Matter Institute at the University of British Columbia were also collaborators on the paper, along with researchers at The Flatiron Institute in New York; the latter provided calculations that demonstrated the change in the Fermi surface.
The Schoop Lab had previously studied two alternate materials using these ultrafast laser techniques, and saw no comparable results. What they saw with the third material, the ZrSiTe, surprised them.
“What started all of this is that we saw what we call these huge coherent phonons in th response that we didn’t see in the other two materials,” said Kirby. “And we only see one – what’s called a phonon mode – in this response. We saw no changes to that as we increased the intensity of the pulse of light. And that sort of tells us that the symmetry of the crystal isn’t changing.”
The Schoop Lab reached out for corroboration to Lukas Muechler, a recent graduate student with the Car Group, now a postdoctoral researcher with the Flatiron Institute who will be joining Penn State Chemistry this summer.
Muechler’s subsequent calculations predicted “very clearly” the change in the material’s Fermi surface. However, the calculations did not accurately demonstrate whether or not there was a symmetry change in the crystal structure of the ZrSiTe sample; the absence of this symmetry change would qualify the transition as an LT.
“A very exciting possibility would be to realize states of matter that usually would not occur in the materials in equilibrium,” said Muechler. “Ideally, one could then control the transitions between these very different states, which would allow us to switch and control the properties of electrons in materials on ultrafast timescales.
“However, there have not been that many experimental observations of transitions between states, which is why we were very excited about the possibility that something like that might happen in ZrSiTe.”
Read the full paper in Physical Review B here: https://journals.aps.org/prb/abstract/10.1103/PhysRevB.103.205138
This work was supported by the National Science Foundation through the Princeton Center for Complex Materials, a Materials Research Science and Engineering Center (DMR-2011750) by Princeton University through the Princeton Catalysis Initiative; and by the Gordon and Betty Moore Foundation (grant GBMF9064). The Flatiron Institute is a division of the Simons Foundation.
