MacMillan Lab’s couple-close advance “makes the impossible, possible”
Nobody knew how to do it. It hadn’t been done. Scientists were skeptical that it could be done. If there is a template for the kind of research that appeals most to the MacMillan Lab, this is it.
A new Science paper advancing their couple-close platform to accommodate a broader range of aromatic rings provides a point of evidence.
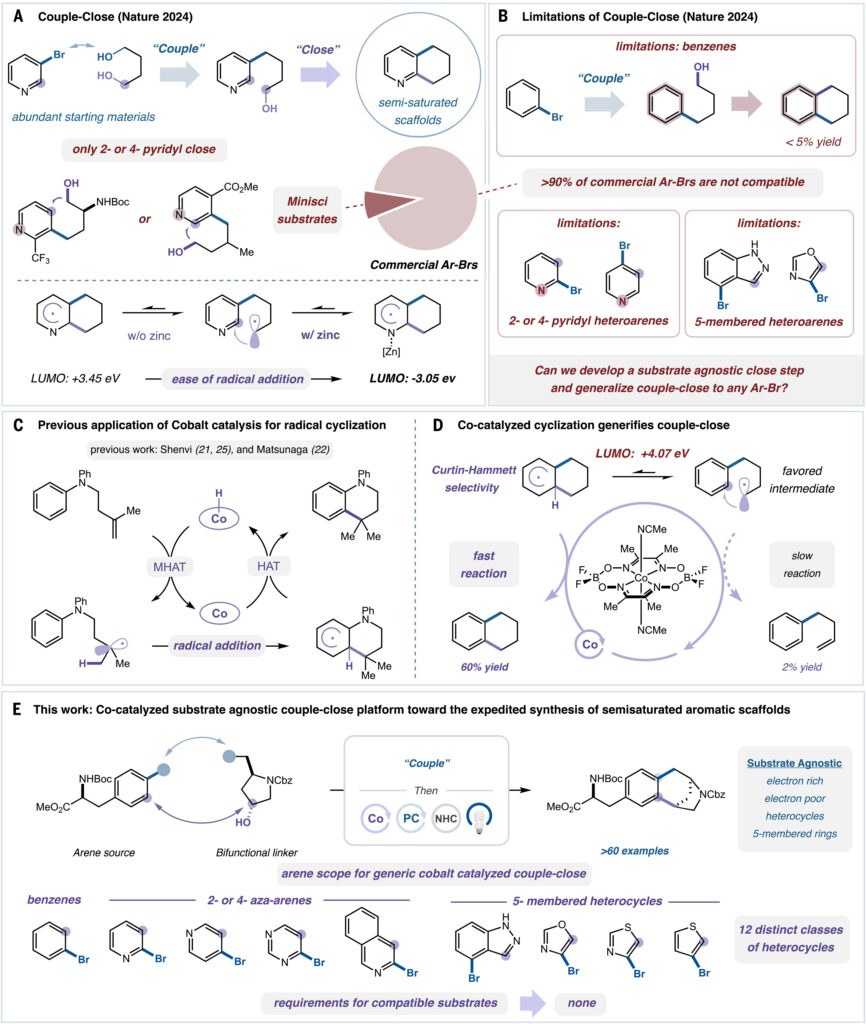
It builds on the couple-close paradigm introduced by the lab two years ago after debate about whether it was feasible to bring two simple fragments together and form a fused ring system in one pot. MacMillan researchers proved it was. Then they pursued the next step: a generalized approach for the synthesis of semisaturated cyclic scaffolds that uses cobalt catalysis as a way to, analogously, close off a loose end.
The result, Couple-close: Unified approach to semisaturated cyclic scaffolds, was recently published in Science.
This NIH-funded research lays out a simple, modular strategy for the synthesis of semisaturated scaffolds, exceptionally valuable motifs in drug discovery. This is significant because these scaffolds have in the past been extremely challenging and labor intensive to build.
Graphical illustration from the journal, Science.
“The best part is you can now make these really cool drug-like molecules rapidly from simple starting materials. It’s a two-step process. You couple and then you close. And it works for all aromatic rings,” said David MacMillan, the James S. McDonnell Distinguished University Professor of Chemistry. “This strategy allows you to build these unique molecules with properties that make them more successful in the clinic. It also gives pharma a way to make them that before might have taken a month or two. Now, they can do it in less than two days.
“Not only can you make one. You can make 20. You can make 100. If you can make it robustly in a day and you can make lots of them, that takes the impossible and makes it possible.”
The key to the enhanced strategy lies in the use of a cobalt-catalyzed dehydrogenation event that enables a radical cyclization to complete the transformation.
“This is the first time group has used cobalt for catalysis. While this particular cobalt catalyst is well known to do hydrogen atom transfer (HAT), a process that our reaction also exploits, it has been rarely applied in the radical cyclization onto aromatic rings.” said William Zhao, a graduate student in the MacMillan Lab and second author on the paper.
“Through this strategy, we can make these fused ring systems where one of them is flat and the other is 3D, and that’s what makes it interesting and unique,” Zhao added. “We would like to see three-dimensionality in drug candidates as that improves binding and solubility in biological systems, but we also want some two-dimensionality in the form of flat aromatic rings because that can also be crucial for structural and conformational rigidity, as well as other interactions with biological systems like pi-stacking.”

William Zhao, a graduate student in the MacMillan Lab and second author on the Science paper.
“These semisaturated scaffolds are the sweet spot where you have the good of the 2D and the good of the 3D. They are challenging to make but highly valuable motifs in agro- and medicinal chemistry.”
A group meeting spurs an idea
Building out these molecules presented challenges right from the start because they can require, among other things, lengthy de novo synthesis. But MacMillan was confident that a little creative thinking could overcome those challenges.
“The idea was, if we can couple one end onto the aromatic and then make the other end ‘close,’ that would be wonderful,” said MacMillan. “But that’s not something we knew how to do in chemistry. So for me, it was a really interesting question. We went out and we did it in context of these specific aromatic rings called pyridines. That paper was published in Nature two years ago.
“But then we were thinking, what if you do it for any aromatic ring in the world? That’s a much bigger thing.”
MacMillan threw the problem out to his group at their weekly meeting. That’s when Jiaxin Xie, a postdoc in the lab at the time, came up with an idea to use cobalt catalysis in a way that expands and generalizes the couple-close paradigm to all aromatic rings, even on electronically unbiased arenes, in a substrate-agnostic manner.
“David envisioned the couple-close strategy to truly generalize the synthesis of semisaturated cyclic scaffolds that address medicinal chemists’ unmet needs,” said Xie. “The major challenge lies in the ‘close’ step. In the close process, traditional strategy involves three steps: radical addition, oxidation, deprotonation-rearomatization.
“So to quickly funnel the shorter lived dearomatized radical intermediate to productive rearomatization under polarity mismatched cases, such as benzenes, a direct hydrogen atom abstraction as a single elementary step would be the ideal mechanistic approach. That should eliminate the unnecessary complications involved in the traditional two-step oxidative rearomatization strategy.
“As for the cobalt catalyst we used for the dehydrogenation process in this work, it has been well-studied and well-understood for HAT processes to generate unsaturation from alkyl radicals – the critical mechanistic aspect of our design,” said Xie. “But it has not been used for dehydrogenative radical cyclization to arenes, which is our opportunity to establish this valuable ‘couple-close’ platform that is truly general.”
The platform can be applied to a broad range of arenes with a variety of electronic properties that are not accessible in the 2024 Nature paper, explained Xie, now a senior scientist at Merck in Boston.
MacMillan said simply, “It was an incredible idea.”
Couple-close: Unified approach to semisaturated cyclic scaffolds was authored by Jiaxin Xie, William Zhao, Johnny Wang, William Lyon, Noriyuki Takanashi, Alice Long, Taylor Sodano, Christopher Kelly, Marian Bryan, and David MacMillan. This research was supported in part by the National Institute of General Medical Sciences (NIGMS) of the National Institutes of Health (grant R35GM134897).
