Scholes Lab: Going quantum to unlock plants’ secrets
When it comes to green living, nobody does it better than plants. When plants convert light into fuel through photosynthesis, not a single particle of light is wasted. If we could unlock plants’ secrets, we might be able to perfect the design of light harvesting in solar cells.
Gregory Scholes, Princeton’s William S. Tod Professor of Chemistry, suspects that the key to plants’ efficiency stems from their ability to harness quantum physics, the unintuitive behaviors of very small particles. In 2010, he led a team that demonstrated quantum effects in marine algae.
But the finding was not without controversy. Quantum behaviors usually reveal themselves at extremely low temperatures isolated from real-world disturbances, raising questions as to whether these quantum states can survive the warm and wet conditions of life.
So Scholes and his team decided to probe quantum behavior in one of the simplest known chemical reactions, the transfer of a hydrogen atom from one part of a molecule to another. If their experiments work out, the researchers could rewrite our understanding of how chemical reactions occur.
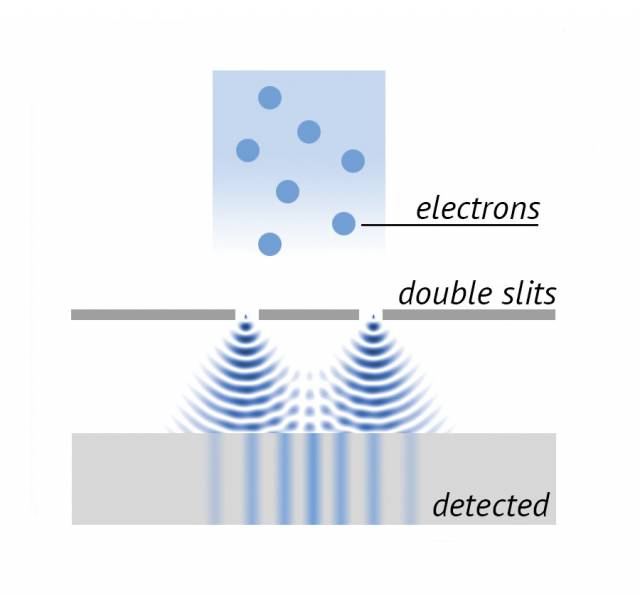
Image courtesy of the researchers
At the heart of quantum theory is the idea that matter can behave both like particles and like waves. If we fire particles at a barrier containing two slits, classical physics predicts that the particles will land in two piles, one behind either slit. In contrast, quantum mechanics predicts that each particle will act like spread-out waves and pass through both slits (see figure), where the intensity at any point can either add together or cancel itself out. …
Read the full article here:
https://www.princeton.edu/news/2019/01/15/going-quantum-unlock-plants-secrets
